Classification of Animals
- Classify into: Unicellular / Multicellular-Invertebrates / Multicellular-Vertebrates:
Earthworm, Snake, Frog, Butterfly, Amoeba, Bacteria, Shark, Ant, Human, Penguin - Give examples of animals which have many legs but are not insects.
- Which vertebrate animal can breathe by moist skin?
- Classify into Fish/ Bird/ Amphibian/ Reptile/ Mammal –
Dolphin, Turtle, Frog, Snake, Penguin, Dinosaur, Duck, Tiger, Shark, Whale
Activity
Find and observe insects in your surroundings. Take their photos. Post only the photos which you have taken yourself.
Find a spider and observe it. Is a spider an insect?
VIDEO
Observe the Chameleon changing colours.
Is a chameleon a vertebrate or an invertebrate? How does it eat food? Is it a reptile or an amphibian? What is the difference between them?
Identify and classify the animal. Identify its property.
Octopus escaping through a small hole
Octopus has soft body and no bones. So it can squeeze from a small hole. It is an invertebrate.
Answer: Chameleon is a vertebrate – reptile. It eats with its long and coiled tongue. Difference – Amphibians can breathe on land as well as inside water. Reptiles can only breathe on land.
Metals and Non-metals
- Classify into Metals/ Non Metals – Iron, Sodium, Sulphur, Potassium, Phosphorus, Calcium, Carbon, Chlorine, Oxygen, Magnesium, Tungsten
- Give an example of a liquid metal and a liquid non-metal.
- What is an alloy? Give 2 examples.
- What is allotropy? Write allotropes of phosphorus and carbon.
- Name 2 inert metals and 2 inert Non-metals.
# Activity
Try to observe and identify objects made of metal or alloys at your home. If you can find five or more such objects of different materials – place them together, name them properly and click a single photo. (Send only ONE photo with all objects named correctly)
Observe the videos –
- Soft and Strong (Reactive Metals)
We have studied about strong (very reactive) metals at our Homibhabha class. Write the names and properties of these metals. Which is the most reactive metal? - Gallium Spoon put in hot tea
What is the spoon made of? What happens to it? - Liquid bromine reacting with aluminium.
Is bromine a metal or non-metal?
Metal Samples
Try to identify the samples seen in our zoom class from this photo.

Answers:
- Zinc cube
- Aluminium cube
- Lead cube
- Copper cube
- Brass cube (Alloy of copper + zinc)
- Brass vessel (same alloy as above)
- Mercury in bottle
- Fools Gold (Iron pyrite – shines like gold, so many attempts were made to extract gold from it)
- Silver (Manohardas Narayandas shuddh chandi 😀) Observe black layer of silver sulphide at the edges.
- Soldering wire
- Nichrome wire (Nickel + Chromium)
Properties of Matter
- Explain in short (one-line): Law of conservation of mass, Sublimation, Malleability
- Amul butter and Water – Which is denser? Do the experiment and find out.
- Find out with experiments – Which vegetables are denser than water?
- Conduct any experiment which demonstrates inertia.
- By any method, find out the volume of any object.
If you do any of these activities yourself, then you can post your photo / video.
Activity
Go to market with your parents. Request the shopkeeper to show you 100gm, 500g, 1kg and 2kg mass. (Hint: Try offering him a chocolate!) Hold the weights in your hands and take a feel of them.
Observe an equiarm balance. Find out which ‘arms’ are ‘equal’. Observe and learn how it is used.
Take a 1 litre bottle and fill it up with water. Find out its mass in grams.
Now remove the water and fill up the bottle with sand. Find out its mass.
What can you conclude from this?
If you do the activity you can post your photo too!
(Don’t forget to thank the shopkeeper!)
Digestive System + Changes in Surroundings
- Explain the role of insulin in your own words. If there is any person using blood-sugar testing device in your family, observe how it is used.
- Study the junk food available at your home like chips, biscuits, cold drinks, etc. Find out how many calories you get if you eat one packet. (It will be printed on the packets.)
Also find out how many calories you get from one cup (200ml) milk. Which one will give you more energy – 1 cup milk or 1 packet junk food? - When do we call a change as a physical or chemical change? Explain with examples.
- Experiment: Take 2 identical glasses. Take 1/4 glassful of drinking water in both. Dissolve two spoonfuls of glucose powder in one of them. Feel the glasses by hand and drink both. Do you observe any temperature difference in them? (If you don’t have glucose powder, try with powdered sugar.)
Optional Activities
- Heating Sugar
Do this ONLY in the presence of your parents.
Take some sugar in a spoon with a sufficiently long handle. Heat it on the gas flame. (Parents to ensure the SAFETY). Observe. Do you observe yellowish-brown liquid?
Heat it for some more time. What do you observe?
- Curdling of milk
Take a few spoonfuls of milk in a container. Squeeze some lemon juice into it. Observe the change.
You will see that the protein (white solid) in the milk gets separated. This is a chemical and irreversible change.
Force and Pressure
- What is the law of action and reaction? Give an example.
- Give an example where we want friction. And one example where we don’t want friction.
- What is the difference between mass and weight? Tell your mass and your weight with proper units.
- Does earth attract you due to gravity? Do you attract earth due to gravity? Do you attract your friend due to gravity?
- What is pressure? Do any experiment to show that “Smaller the area, more is the pressure”.
Activity –
Optional Activities:
Try any of these activities seen in class (whichever you can):
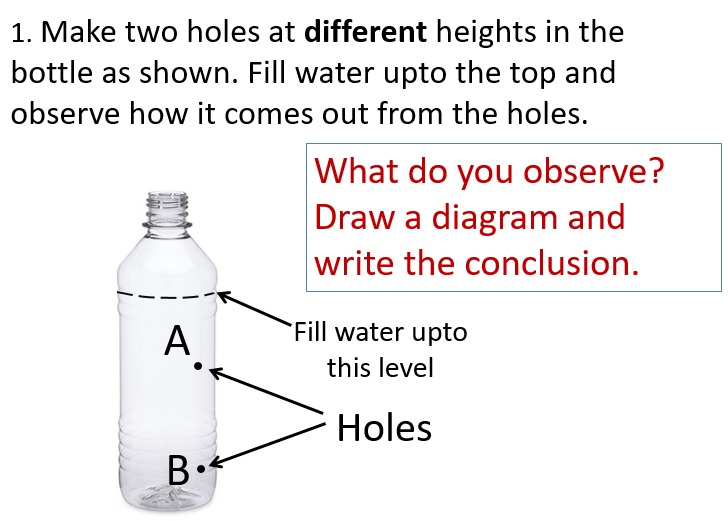
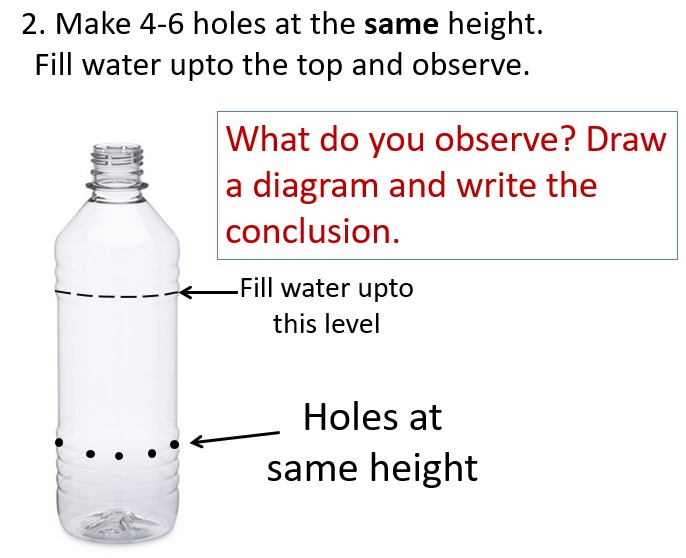
- Make two small holes in a plastic bottle at different heights. Fill the bottle with water and observe the flow of water. What do you observe? What can we conclude from this experiment?
- Make 4-5 holes in a plastic bottle at the same height. Fill the bottle with water and observe the flow of water. What do you observe? What can we conclude from this experiment?
- Fill a glass with water up to the brim. Put a plastic lid on top. Press the lid slightly and invert the glass. Slowly leave the lid and observe. Does the water spill out? Why?
- Try to remove water from a container at some height using the siphon method seen in class.

Classification of plants
- Observe various types of foodgrains, spices, oilseeds, etc used in food preparations in your house. Learn their names. Identify which are monocots and which are dicots.
- Classify the following plants:
First identify if the plant is flowering or non-flowering. If it is flowering, also identify if it is monocot or dicot –
Cauliflower, Algae, Beetroot, rice, cactus, bamboo, mango, coconut.
Answers:
Q1. Some plant products you might have found in your house:
Dicots – All Pulses like moong, matki, chawli, green gram, lentil, soyabean, dalchini, clove, oilseeds (groundnut, til, sesame,flax, mustard, cashenut, almond),
Most vegetable plants like cabbage, carrot, tomato, brinjan, bhendi, palak, methi, onion, potato, etc are also dicots.
Monocots – rice, wheat, jowar, ragi, nachni, sugarcane, elaichi, banana, supari (betelnut), maize
Q2. Classify the following plants:
Non flowering plant – Algae
Flowering Plants:
Monocots – rice, bamboo, coconut.
Dicots – Cauliflower, Beetroot, cactus, mango
Respiration and Circulation
Measure your pulse rate = Number of heartbeats in one minute.
Also measure your rate of breathing = How many times you breathe in one minute.
Jump up and down continuously for 1 minute.
Now measure your pulse rate and breathing rate again.
What do you observe? Explain.
ANSWER:
Generally rate of heart beats is 72 per minute. But it can vary considerably from person to person.
You would have observed that heartbeats as well as breathing rate increase as you start jumping.
As we do some exercise our cells use up more oxygen from the blood and CO2 level in the blood increases. So to supply more oxygen to the body, our breathing rate and heartbeats increase.
Simple Machines
Name and identify the simple machines shown in these images. Make a table with columns as – Name, What is at the centre, Type (as we did in class)
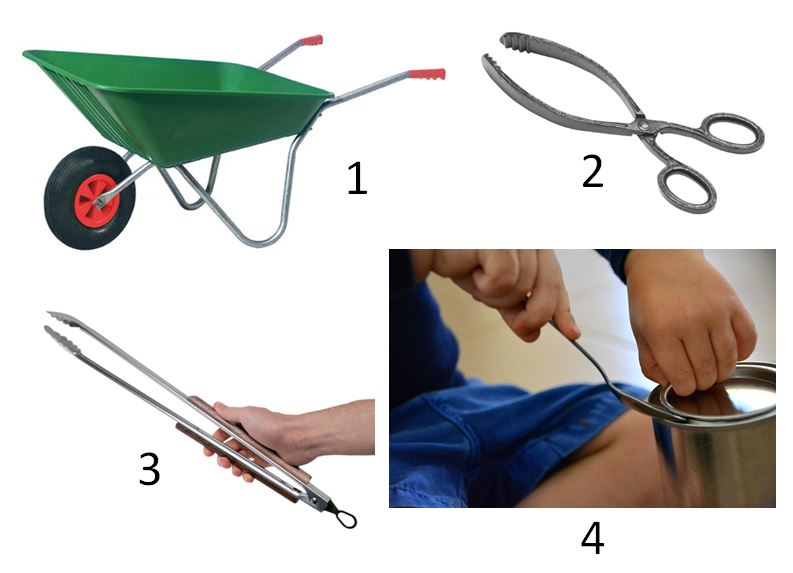

Also identify objects around you that make use of simple machines. If you find 4 or more, you can post a photo with labels.
ANSWERS
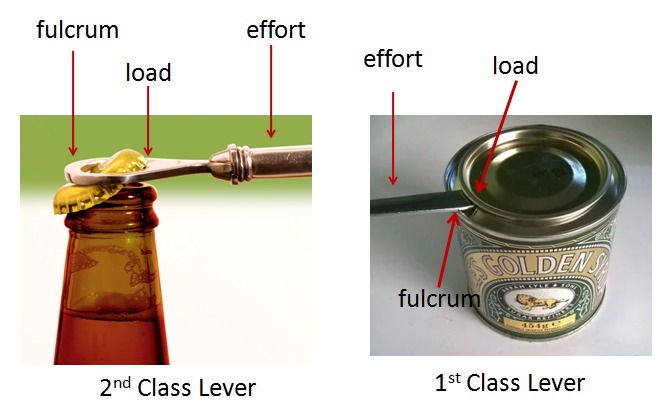
- Wheelbarrow – Lever Type 2
- Scissor shaped tongs – Lever Type 1
- Coal Tongs – Lever Type 3
- Spoon used to open a can lid – Lever Type 1
- Pan Balance – Lever Type 1
- Bottle Opener – Lever Type 2
- Pulley – Not a Lever
- Oars for rowing – Lever Type 1
- Claw of hammer used to extract a nail – Lever Type 1
Some explanations shown in the images below –