These are notes from our online Zoom class for 9th Std Homi Bhabha Exam
Topic: Metals and Non-Metals
Observe the samples, record the names and note the properties.

Observations
- Metals: Aluminium, lead, Cu, Zn, iron, Bismuth, silver, gold, mercury (liquid)
- Black silver sulphide, and greenish colour copper carbonate
- Density of lead is more than aluminium.
- Lead is relatively soft and can draw a line on a paper.
- Alloys: Nichrome, Solder alloy, Brass
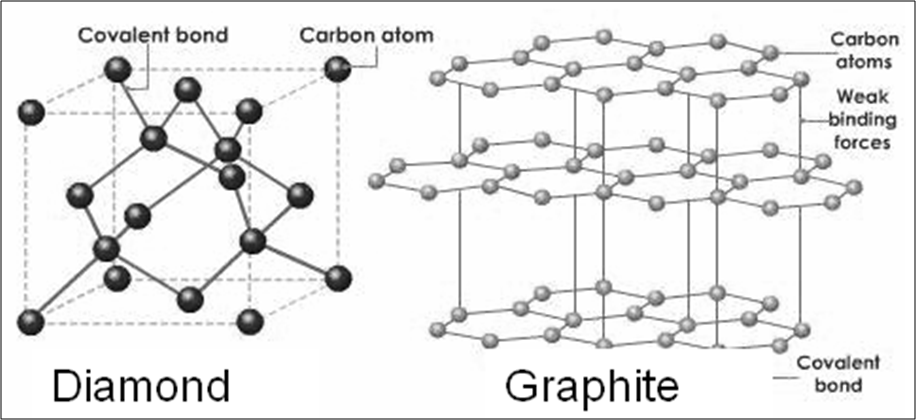
- Non metals: Coal, graphite, diamond (all are allotropes of Carbon), Sulphur, red phosphorus on matchbox
- Metals are solid and shining.
- Metals are denser, strong. Metals are sonorous.
- Metals are ductile and malleable

Properties of Metals
- Sonorous
- Conductor of heat and electricity
- Metals tend to donate the electron and form + ions
- Non metal oxides are mostly acidic. (except CO, NO, N2O, H2O)
- Metal oxides and hydroxides are mostly basic. Example MgO
- Chromic oxide Cr2O3 and metalloid antimony oxide (Sb2O3) are acidic
- Zn and Al oxides are amphoteric:
- Al2O3 + 6HCl → 2AlCl3 + 3H2O
- Al2O3 + 2NaOH → Na2Al2O4 + H2O
Properties of metals
- Reactivity, inertness
Reactivity series: K, Na, Ca, Mg, Al, Zn, Fe, Pb, (H), Cu, Ag, Pt, Au
- Cu(SO4) + Fe —-> Fe(SO4) + Cu
- Fe(SO4) + Cu —-> ?
Gold is inert! (Remember the video – Gold is the BEST!)
Gold dissolves in freshly prepared Aqua Regia (HNO3+3 HCl)

2. Oxides
Metals react with oxygen to form oxides.
(Iron rusts over days, while potassium burns in seconds)
Examples
- 4 Na + O2 → 2 Na2O (sodium oxide)
- 2 Ca + O2 → 2 CaO (calcium oxide)
- 4 Al + 3 O2 → 2 Al2O3 (aluminium oxide)
3. Reaction with water
- Water is H-OH
- Metals which are reactive than hydrogen, form hydroxide and hydrogen gas is evolved.
- 2Na + 2H2O –> 2NaOH + H2 (Explosive reaction)
- Fe has reversible reaction with water.
- 3Fe + 4H2O –> Fe3O4 + 4H2
- Rusting of iron (in presence of oxygen and water)
4Fe + 3O2 + H2O –> 2Fe2O3. nH2O
(rusting is faster if water contains acids or electrolytes or if iron is impure) - Mg reacts only with steam
- Aluminium shows no reaction with water. Pb, Cu, do not react with water
Strong alkali metals
Recall the videos – - Explosive reaction of Lithium, Sodium, potassium, rubidium, Caesium
- Rubidium, caesium put in a tub!
4. Reaction with acids
- K + 2HCl –>2KCl + H2
- Zn + 2HCl –> ZnCl2+ H2
- Fe + H2SO4 –> FeSO4 + H2
- Pb + [conc] HCl –> PbCl2 + H2
- Platinum (Pt) and Gold (Au) don’t react even with concentrated acids. (Remember, Gold is the Best!)
5. Reducing the metals: (Getting pure metals from their oxides.)
- Fe2O3 + 3CO –> 2Fe + 3CO2
- PbO + C –> Pb + CO
- ZnO + C –> Zn + CO
- CuO + H2 –> Cu + H2O
Less reactive metals can be reduced by thermal decomposition - 2HgO –> 2Hg + O2
- 2Ag2O –> 4Ag + O2
- Pt, Au do not form oxides.
6. Physical properties of metals
- Liquid metals at room temperature (Examples: Mercury, Gallium, rubidium, Caesium)
- Alloys
Alloy is a mixture of two or more metals. It is a physical change. But it changes the properties of the metals drastically.- Brass = Copper + zinc
Cu and brass require tinning
- Mercury alloys are called amalgams. Mercury can dissolve gold, sodium, zinc forming amalgam.
- Duralumin, magnalium are alloys of aluminium with copper and magnesium, used to make aircraft parts
- Al powder + Fe2O3 is used as thermite mixture for welding iron
- Brass = Copper + zinc
- Coatings: tinning, galvanizing: Zinc coating on iron
- Heat treatment:
- Hardening or quenching: it is a process in which steel is heated till it becomes red hot to temperatures above 800 0C. Then it is suddenly plunged into cold water or oil. Due to this hardness increases. But steel also becomes more brittle.
- Annealing or tempering- it is a process in which hardened steel is heated at temperatures between 220 0C to 300 0C. Then it is cooled very slowly over a period of four to five hours. This increases toughness of the steel. It retains the hardness but reduces the brittleness.
Metallurgy
- metal ores
- ‘matrix’ or gangue’: rocky impurities associated with ore (like silica SiO2)
- Flux: chemical added to get rid of matrix
- Flux + matrix—> ‘slag’ which is separated from metal easily
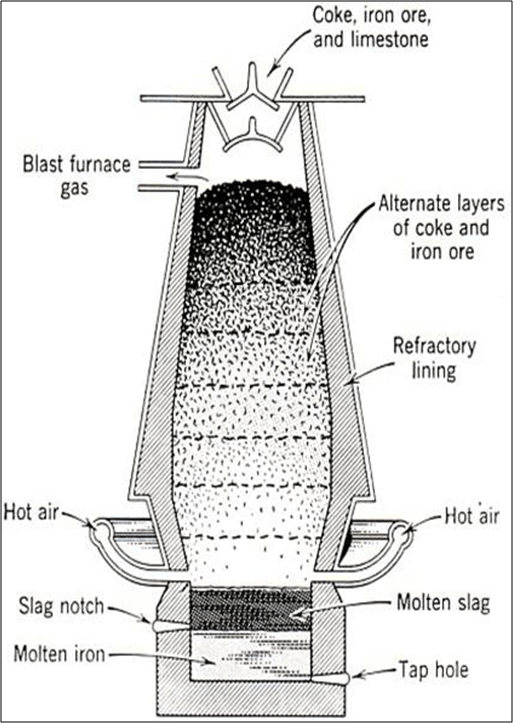
Iron:
- Mining
- Concentration of ore
- Furnace

- Concentration of ore
- Magnetic separation
- Gravity separation
- Froth flotation
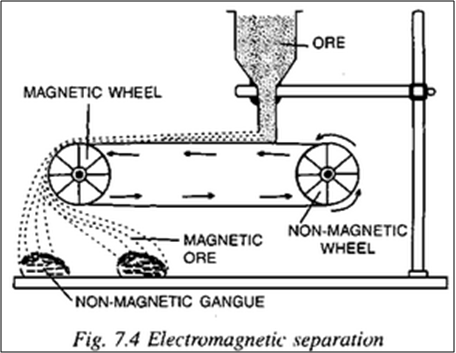
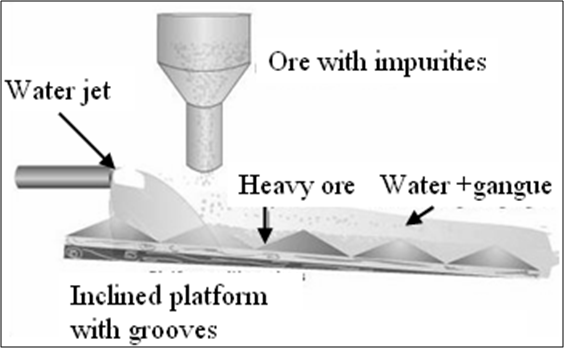
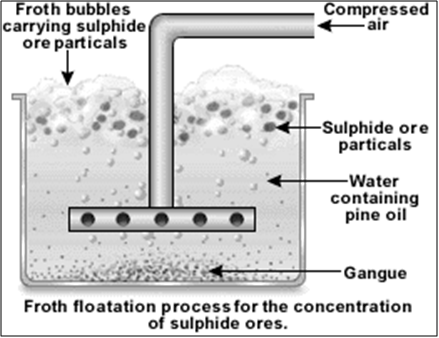
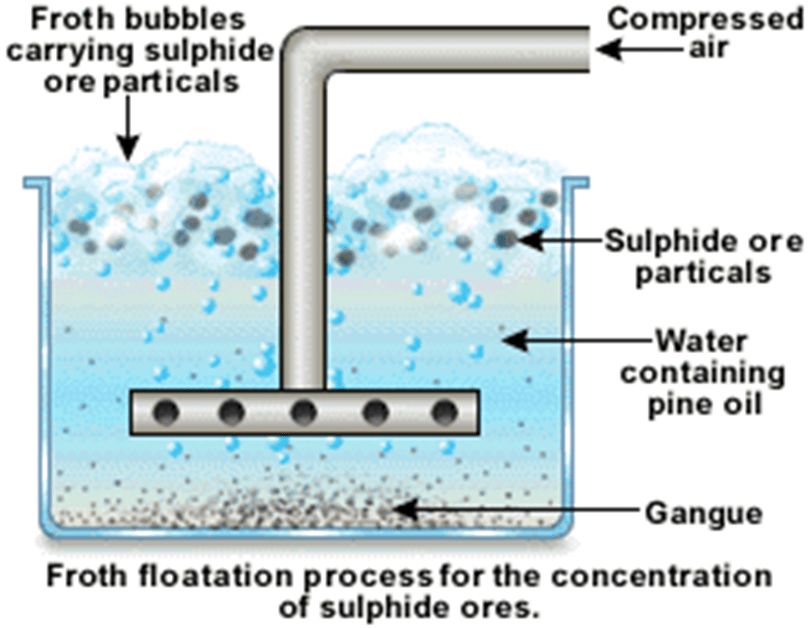

Extraction of iron: Blast Furnace
1. C + O2 → CO2↑ + heat
2. CO2 + C → 2CO
3. CaCO3 → CaO + CO2
4. CaO + SiO2 → CaSiO3 (slag)
5. Fe2O3 + 3 CO → 2Fe + 3CO2


- Metal oxides – Zincite (ZnO), Heamatite (Fe2O3), Bauxite (Al2O3.2H2O), Cuprite (Cu2O)
- Carbonates- Marble (CaCO3), Calamine (ZnCO3), siderite (FeCO3), Magnesite(MgCO3)
- Halides – Fulorspar (CuF2), Cryolite (Na3AlF6), Horn silver(AgCl), rock salt (NaCl)
- Sulphides – Zinc blende (ZnS), Galena(PbS), Iron pyrite(FeS2), Cinnabar (Hgs)
- Sulphates – anglesite (PbSO4), baryte (BaSO4), Gypsum (CaSO4.2H2O), Epsom salt (MgSO4.7H2O)
Some uses of metals
- Cooking vessels are made up of metals like steel and alluminium. As they are good conductors of heat.
- Electric wires are made up of mostly copper, sometimes of aluminium
- Our bones contain calcium carbonate and calcium phosphate
- Blood haemoglobin contains iron (Fe)
- Nickel is used to make coins.
- Tungsten is used to make filaments of electric bulbs.
- Titanium compounds are used in paints.
- Aluminium foil
- Silver bromide can have light sensitive reactions. It is used in photography

Radioactive elements
• Uranium, Thorium, Radium, Plutonium
• Radium was discovered from ores of pitchblende by Madam Curie (Noble prize).
• Thorium is found in Kerala.
• Uranium is useful for atomic energy and atom bomb
Non-Metals
- Non-metals do not have luster (shiny appearance), are bad conductress of heat and electricity. Not ductile, not malleable. Have low melting point, low boiling point, not sonorous (do not produce sound).
- Hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, iodine, carbon, phosphorus, sulphur, are non-metals.
Solid: carbon, iodine, Sulphur, Phosphorus
Liquid: Bromine
Gas: Oxygen, hydrogen, nitrogen, chlorine, argon helium etc.

Very reactive non metals:
Fluorine, chlorine, bromine, iodine
Oxygen is also reactive.
Helium, neon, argon, krypton, xenon are inert gases
Most of the gaseous non metals form molecules by sharing electrons. For example two oxygen atoms form oxygen molecule O2.

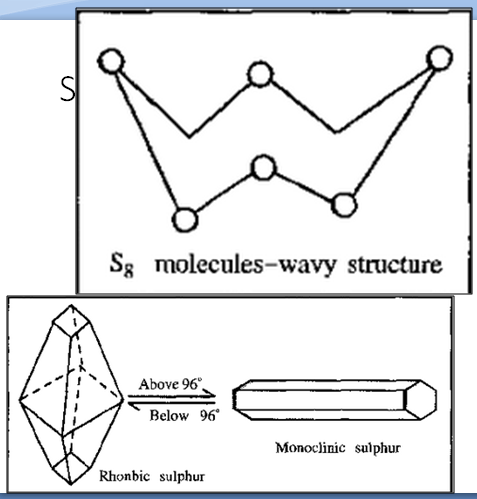
Allotropes of Sulphur-
Sulphur is usually found in form of yellow powder. It can also form needle shaped crystal structure. Sulphur is poisonous. Sulphur can burn with blue flame forming sulphur di oxide gas. (SO2) it causes pollution. And can cause acid rain. Sulphur is also used in medicines like sulpha drugs. Onion, garlic, eggs contains sulphur.

Allotropes of Phosphorus-
phosphorus has two allotropes red phosphorus and yellow phosphorus. Yellow phosphorus burns at very low temperature (even at room temperature). So it is stored under water. Red phosphorus is used in making safety matches. (The special surface made on the side of the box contains red phosphorus and not on the match sticks.)

• Phosphorite Ca3(PO4)2, Chlorapetite 3Ca3(PO4)2.CaCl2, Fluorapatite 3Ca3(PO4)2.CaF2
• Has garlic like odor.
• Catches fire at 300C. Hence kept under water. It shows phosphorescence
• Red phosphorus is produced by heating yellow phosphorous in inert atmosphere of CO2 or N2. Iodine is used as catalyst.
• Red phosphorous does not show phosphorescence
White phosphorous can react with hot concentrated sodium hydroxide to form phosphine gas and sodium hypophosphite.
P4 + 3NaOH + 3 H2O → PH3 + 3NaH2PO2
Metalloids-
Some elements shows properties of both metals and non metals. They are called metalloids. Eg. Silicon, germanium, arsenic, Boron, antimony Silicon is present in sand as silicon dioxide.(SiO2)
Silicon and germanium are semiconductors. Their electrical condicivity can be changed by adding small amount of other elements. They are used in computers, mobiles, calculators

Silicon
•grey colored metalloid. Semiconductor.
•sand is silicon dioxide SiO2.
•present in flint, quartz, opal, mica.
•SiC (silicon carbide) is a very hard substance used to make cutting tools. It has high melting point 1410 0C.
•Sodium silicate Na2SiO3 is called water glass. In this solution different salts like copper sulphate, ferrous sulphate are dropped. After few hours they form tube like structures which look like artificial plants. This is called silica garden.
