This is a compilation of the points we studied in our Homi Bhabha Exam Online Class.
Topic: Carbon Chemistry
Elemental carbon
- Symbol: C
- Atomic number : 6
- Electronic configuration (2,4)
- Atomic mass: 12 (most common isotope)
Carbon: allotropes
• Crystalline: Diamond, graphite
• Amorphous: coal, coke, fullerenes

| Type of Coal | Carbon content | Commonly known as |
| Peat | 11% – 60% | – |
| Lignite | 60-70% | Soft coal / brown coal |
| Bituminous | 70-90% | Household coal |
| Anthracite | 96% | Pure, Hard coal |

Carbon is present in all known life forms. In the human body, carbon is the second most abundant element by mass (about 18.5%) after oxygen. There are an immense number of distinct compounds that contain carbon atoms. Carbon has the ability to form very long chains of interconnecting C-C bonds. This property is called catenation. Carbon-carbon bonds are strong, and stable. This property allows carbon to form an almost infinite number of compounds. Study of these compounds is done in a branch called ‘organic chemistry’.
Western alchemists believed that, certain compounds could only be synthesized from classical elements (Earth, Water, Air and Fire) by action of a “life-force” possessed only by organisms. These compounds were called “organic” compounds. Whereas “inorganic” compounds could be obtained from the elements by chemical manipulation.
Friedrich Wöhler synthesized oxalic acid, a compound known to occur only in living organisms, from cyanogens (a chemical – (CN)2). A more decisive experiment was synthesis of urea from the inorganic salts potassium cyanate and ammonium sulfate (1828). Urea had long been considered to be an “organic” compound as it was known to occur only in the urine of living organisms. Later on many such increasingly complex “organic” substances were produced from “inorganic” ones without the involvement of any living organism.
The modern meaning of “organic compound” is any one of them that contains a significant amount of carbon.
International Union of Pure and Applied Chemistry (IUPAC)
The main idea of IUPAC nomenclature is that every compound has one and only one name, and every name corresponds to only one structure of molecules

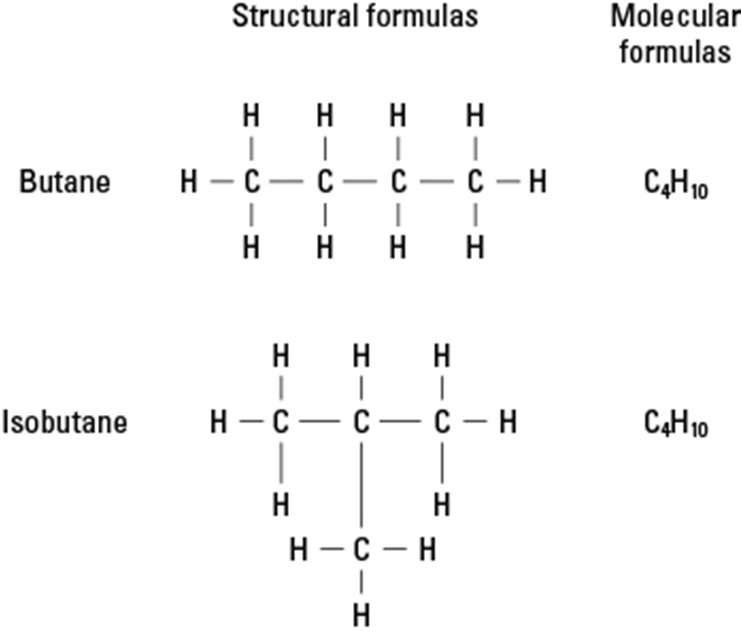
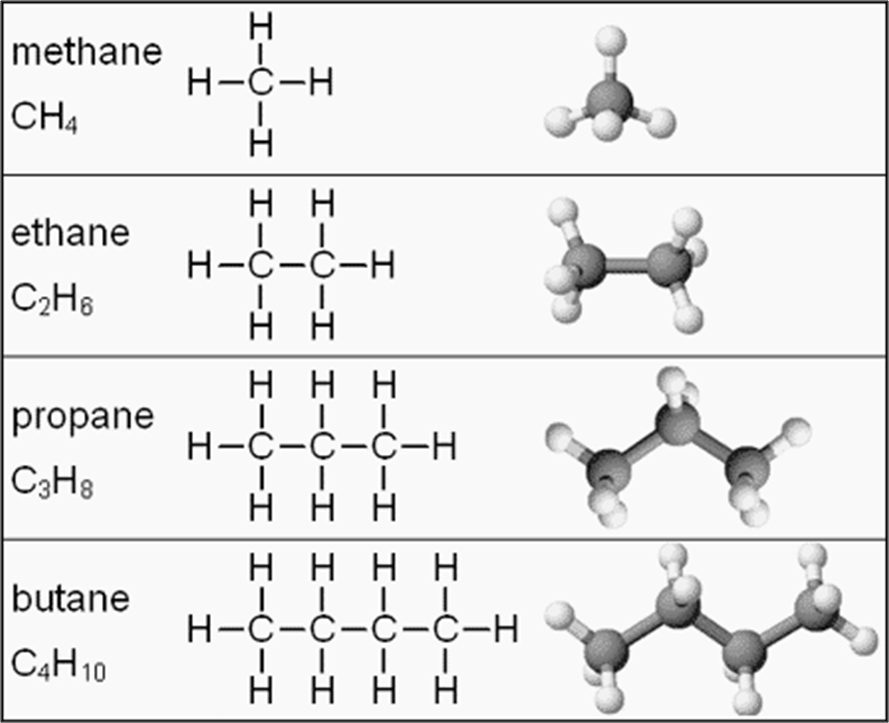
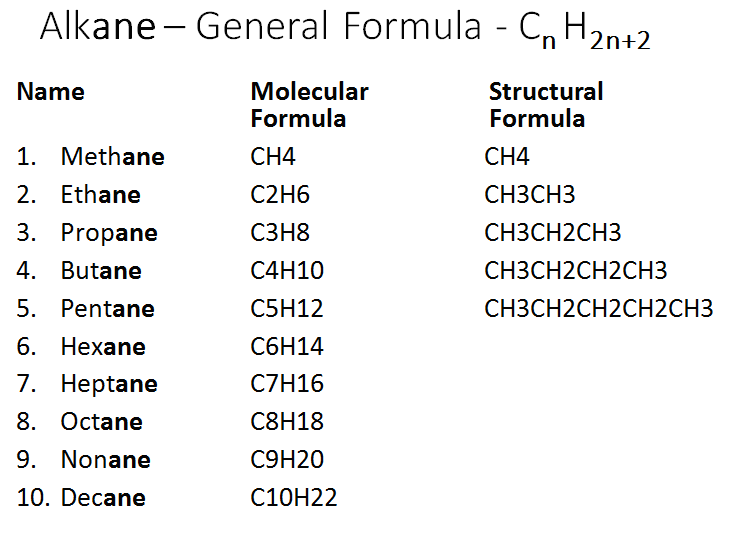
Alkanes – Single bond
Burning of alkanes: Higher alkanes have little value and are usually split into lower alkanes by the process called ‘cracking’.
In general, alkanes show a relatively low reactivity, because their C bonds are relatively stable and cannot be easily broken. Alkanes generally do not react to acids and bases. Due to this inertness they are called paraffins (lacking affinity). In crude oil the alkane molecules have remained chemically unchanged for millions of years.
All alkanes react with oxygen in a combustion reaction, although they become increasingly difficult to ignite as the number of carbon atoms increases. The combustion is without any smoke.
The most important source of alkanes is natural gas and crude oil. Alkanes are separated in an oil refinery by fractional distillation and processed into many different products.

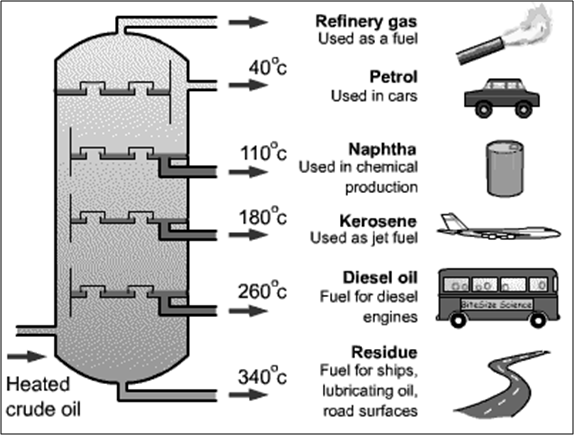
Uses of Alkanes
The applications of a certain alkane can be determined quite well according to the number of carbon atoms. The first four alkanes are used mainly for heating and cooking purposes, and in some countries for electricity generation. Methane and ethane are the main components of natural gas; they are normally stored as gases under pressure. It is, however, easier to transport them as liquids: This requires both compression and cooling of the gas.
Methane is obtained from gobar gas, sewage gas, biogas and coal gas. It is also known as marsh gas as it is naturally formed by bacteria in marshy areas due to decomposition of vegetable matter. Methane can be produced by heating sodium acetate and sodalime. It produces sodium carbonate and methane gas.
CH3COONa + NaOH (+CaO) –> Na2CO3 + CH4
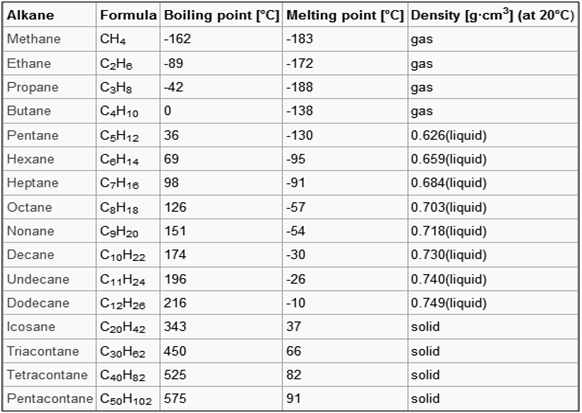
Propane and butane can be liquefied at fairly low pressures, and are well known as liquified petroleum gas (LPG). Propane, for example, is used in the propane gas burner, butane in disposable cigarette lighters.
From pentane to octane the alkanes are reasonably volatile liquids. They are used as fuels in internal combustion engines. Alkanes from hexadecane upwards form the most important components of fuel oil and lubricating oil. As they are hydrophobic in nature, they are anti-corrosive agents.
Many solid alkanes find use as paraffin wax, for example, in candles. Alkanes with a chain length of approximately 35 or more carbon atoms are found in bitumen, used, for example, in road surfacing. However, the higher alkanes have little value and are usually split into lower alkanes by the process called ‘cracking’.
Isomerism –
Alkanes with more than three carbon atoms can be arranged in numerous different ways, forming different structures. This property is called isomerism.
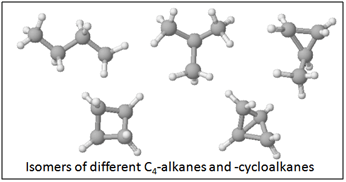
Cycloalkanes: CnH2(n+1-g)
Types of alkanes which have one or more rings of carbon atoms
g = number of rings
(Remember how we checked the formula for 1, 2, 3 rings?)

Alkene – Double Bond – CnH2n
Alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond.
The simplest alkenes, with only one double bond, form a homologous series of hydrocarbons with the general formula CnH2n.
The simplest alkenes, ethene, propene and butene are gases. Linear alkenes of approximately five to sixteen carbons are liquids, and higher alkenes are waxy solids.
| Name | Molecular formula | Structure |
| Ethene | C2H4 | CH2=CH2 |
| Propene | C3H6 | CH2=CH-CH3 |
| 1-butene | C4H8 | CH2=CH-CH2-CH3 |
| 2-butene | C4H8 | CH3-CH=CH-CH3 |
| 1-pentene | C5H10 | CH2=CH-CH2-CH2-CH3 |
| 2-pentene | C5H10 | CH3-CH=CH-CH2-CH3 |
Note
- There is no 3-butene, because the same molecule seen from the other side will be 1-butene
- Similarly there is no 2-propene
- Similarly, there is no 3-pentene and 4-pentene
- But 3-hexene is there!
- How many different octenes can be there?
Polymerization:
Polymerization of alkenes – in this process many alkene molecules come together to form a very long chain called polymer. This is also called Catenation. For example ethylene molecules form polyethylene. Polymers are of high industrial and economical value, such as the plastics polyethylene and polypropylene. Billions of kilograms of these materials are made and used each year.
• Polyethene – most common plastic in use today
• Many kinds of polyethylene are known, with most having the chemical formula (C2H4)n.
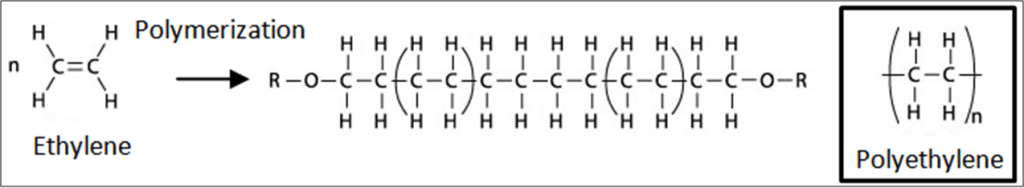



Vegetable oils are unsaturated double bond carbon compounds. They are saturated by addition of hydrogen to make vegetable ghee (Dalda)
Alkyne: Triple bond
| Name | Molecular formula | Structure |
| Ethyne | C2H2 | CHºCH |
| Propyne | C3H4 | CHºC-CH3 |
| 1-butyne | C4H6 | CHºC-CH2-CH3 |
| 2-butyne | C4H6 | CH3-CºC-CH3 |
| 1-pentyne | C5H8 | CHºC-CH2-CH2-CH3 |
| 2-pentyne | C5H8 | CH3-CºC-CH2-CH3 |
Aromatic Hydrocarbons – Generally having alternating double and single bonds between carbons. They have sweet scent (aroma).
(This structure of benzene was really discovered while dreaming!)
Some examples:
Alcohol, Aldehyde, Acid group
- Aldehyde- these are organic compounds which have CHO group attached.
Organic acids – these are organic compounds having carboxyl group (COOH) attached. Formic acid H-COOH, Acetic acid CH3-COOH. Aldehydes and acids are used to produce dyes, esters and other organic compounds.
- Alcohol – Alcohol is obtained by replacing one of the hydrogen from hydrocarbons by hydroxyl (OH) group.
Examples: Methanol (CH3OH), Ethanol (C2H5OH). - Methyl alcohol (methanol) is obtained by heating wood in absence of air. It is colourless, poisonous liquid. It is a good solvent. It has alcoholic odour. It is used as solvent for wood polish, paints, varnishes, it is used to make perfumes. Also used to make methylated spirit.
(Remember the difference we discussed between Methanol and Ethanol?
- Glycerin (Glycerol)



- Ketone – Dimethyl ketone (Acetone) (CH3-CO-CH3) is simplest ketone – Used in nailpaint removers
- Ethers- have general formula R-O-R’. good solvent, and a fuel.
- Esters – have general formula (R-CO-O-R’). commonly used as fragrances and found in essential oils
- Freon (CCl2F2) – it is used in refrigeration and air conditioning as a cooling agent. It is also used to produce insecticide aerosol
- Carbontetrachloride (CCl4) – Carbon tetrachloride is a colour less liquid with sweet smell. It was used as dry cleaning solvent, as a refrigerant and also as fire extinguishers. It is ozone-depleting and a greenhouse gas. Due to its harmful environmental effects its use is reduced now. It is a very good solvent which can dissolve oils, fats and also iodine.
- Chloroform (CHCl3) – Chloroform was once a popular anesthetic. Its vapor depresses the central nervous system of a patient, allowing a doctor to perform various otherwise painful procedures. Due to its toxic effects it is not used now.
- Aerosols

- Polytetrafluoroethylene (PTFE) (C2F4)n
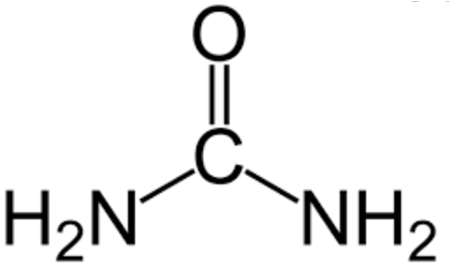
- Urea – CO(NH2)2
- Oxalic acid – COOH-COOH.2H2O

Common source of confusion:
- Ethane: C2H6
- Ethene: C2H4 (Ethylene is trade name – don’t think it is an alkyne because of the ‘y’ in the spelling. It is an Alkene. )
- Ethyne: C2H2 (Acetylene is trade name. It is an Alkyne)


